
The Northside Hospital Cancer Institute Blood Cancer Program combines cutting-edge treatment and the most current clinical trials with the compassionate and supportive care you expect from Northside. Nationally recognized physicians and hematopathologists work alongside experts in our program’s blood bank, stem cell laboratory, immunotherapy program and blood and marrow transplant program.
Types of blood cancers and related disorders treated at Northside Hospital include:
As the largest cancer network across Georgia, our patients do not have to travel far for our powerful combination of cancer expertise, treatment facilities and support resources. We offer on-site state-of-the art diagnostic molecular testing, flow cytometry and dedicated disease site pathologists, and our medical oncologists are national leaders in advanced cancer treatment and research.
Patients with certain forms of blood cancers have access to our nationally recognized Leukemia Program and Blood and Marrow Transplant (BMT) Program at Northside Hospital (NH-BMT). These programs operate with highly experienced and specialty trained physicians and staff. Additionally, bone marrow transplant patients benefit from early access to donor searches and a seamless transition to our Blood & Marrow Transplant Program.
In January 2020, Jack and Becky Miller became empty nesters, with their children in college or with their own families. Jack, a pilot and captain instructor for a national airline, and Becky had their bucket list primed, full of ideas and adventures they were eager to check off...

Northside Hospital Cancer Institute's Cancer Research Program is one of the largest community-based oncology/hematology programs in the nation and is one of the few cancer research programs in the country offering Phase I-IV clinical trials.
Northside Hospital is committed to offering the latest in cancer research, breakthrough procedures, technologies and treatments through collaborations with some of the most respected cancer research institutions and programs in the country.
DISCLAIMER
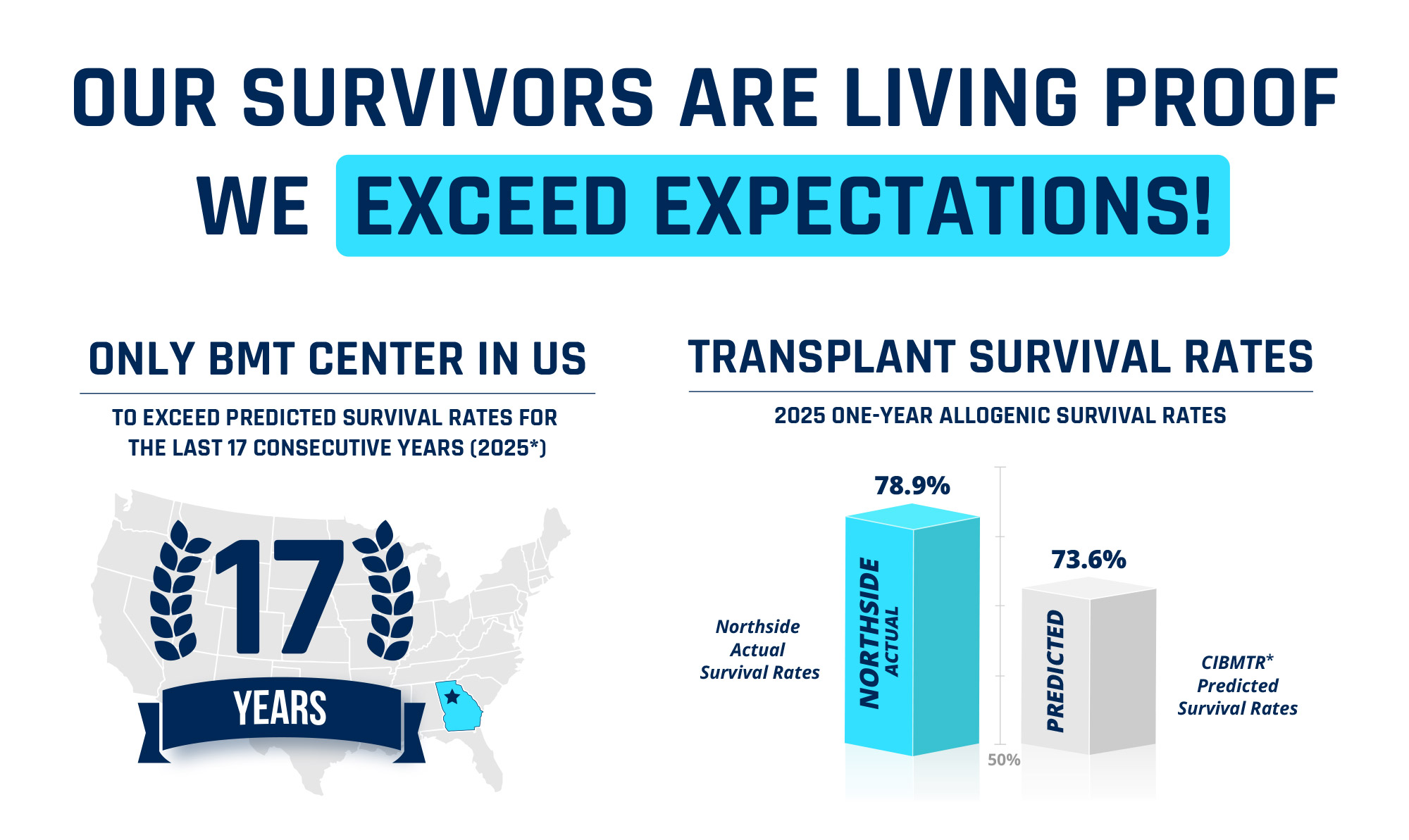
1 2025 Transplant Center Specific Survival Report, December 2025. This survival information includes only patients who received their first allogeneic transplant between January 1, 2021 and December 31, 2023, using unrelated or related donors and who had reported follow-up. Specific transplant center information can be found at nmdp.org.